SUSTech research group makes significant progress in the study of Magnetic Classification Theory
2026-04-23Ambient-pressure Nickelate Superconductors reach new heights
2026-03-13SUSTech Team Reveals Atomic-Scale Mechanism Governing Thermoelectric Transport in Bismuth Telluride
2026-03-06SUSTech team advances the construction of a tunable 2D “Kondo Superlattice”
2026-01-27Researchers make breakthrough in magnetic geometry-induced quantum geometry and nonlinear transport
2025-06-06High entropy alloy nanoparticles (HEA-NPs) show great potential as functional materials. However, thus far, the realized HEAs have been restricted to palettes of similar elements, which greatly hinders the material design, property optimization, and mechanistic exploration for different applications.
Associate Professor Junhao Lin’s research group from the Department of Physics at the Southern University of Science and Technology (SUSTech) and the Guangdong-Hong Kong-Macao Greater Bay Area Quantum Science Center, and Professor Lei Fu’s research group from the School of Chemistry and Molecular Science of Wuhan University, have made a breakthrough in the study of the synthesis and growth mechanism of high entropy alloy nanoparticles (HEA NPs).
The synthesis of HEA generally requires harsh experimental conditions such as high temperature and high pressure. In order to overcome this problem, the researchers developed a new synthesis method of liquid metal gallium (Ga) assisted HEA NPs, which can obtain HEA NPs with uniform and mutual solubility of up to 17 elements under relatively mild experimental conditions. The growth mechanism of HEA NPs was studied by in-situ transmission electron microscopy.
Their findings, entitled “Liquid metal for high entropy alloy nanoparticles synthesis”, were published in the top international journal Nature.
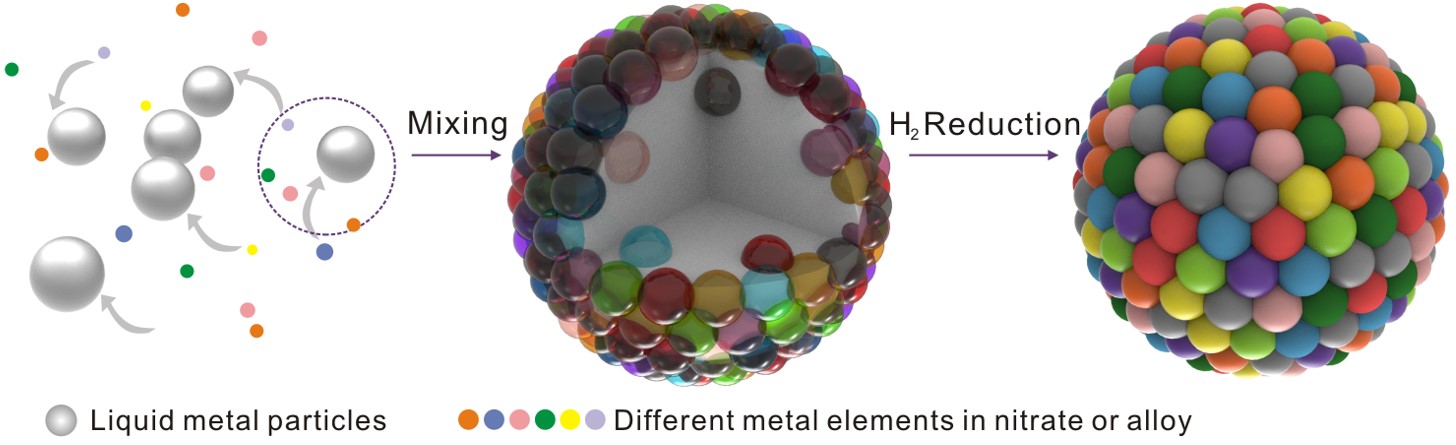
Figure 1. Schematic diagram of liquid-metal assisted synthesis of HEA NPs
HEA NPs usually contain at least five metal elements. High specific surface area, strong synergistic effect of elements, customizable element components, and large lattice distortion make them an ideal reaction platform for a variety of surface reactions. The great differences in the chemical and physical properties of many mixed elements result in immiscibility between elements during alloying. Although the high entropy state facilitates uniform mixing between different elements, this process requires not only extremely high reaction temperature, but also a quenching process to stabilize the high entropy state at room temperature. In addition to the extreme reaction conditions, the reported composition of HEA NPs is limited to the elements with similar properties, which significantly hinders the exploration of HEA NPs in the fields of material design, property optimization, and specific functionalization.
In this work, the team reported that liquid metal has unique advantages in the synthesis of HEA NPs: on the one hand, its mixing enthalpy with other metal elements is negative, which can provide stable thermodynamic conditions for the mixing of multiple metal elements; On the other hand, the fluidity of liquid metal can provide sufficient kinetic conditions for mixing between different elements, and HEA NPs with wide melting point range (303-3683 K) and wide atomic radius range (1.24-1.97 A) can be synthesized under mild reaction conditions. They further achieved precise fabrication of nanoparticles composed of different elements by adjusting the enthalpy of mixing.
HEA NPs have high surface activity, which are easily oxidized by air and adsorb hydrocarbons in the environment, which seriously restricts the characterization of samples and physical property tests. The glove box interconnection system (GIS) with full inert atmosphere protection built by Prof. Lin’s group (Fig. 2) realized full inert atmosphere protection from sample growth, transfer, post-processing, atomic structure characterization, and physical property testing, which greatly reduced the surface oxidation and pollution of HEA NPs. It has the unique advantage of studying HEA NPs with high surface activity.

Figure 2. Schematic diagram of glove box interconnection system (a) and the related photograph (b)
The researchers combined the liquid metal-assisted growth of HEA NPs with the GIS to achieve elemental distribution and atomic STEM structure characterization of intrinsic samples. As shown in Fig. 3a, HEA NPs containing up to 11 different elements (CuPdNiMnAlInGaRhPtCoMg) were confirmed. The EDS distribution diagram proves that each component element is uniformly distributed in the HEA NPs. The corresponding atomic resolution scanning transmission high angle annular dark field images (HAADF-STEM) and fast Fourier transform (FFT) images show that HEA NPs obtained under relatively mild reaction conditions have high-quality crystallinity.
Through the auxiliary synthesis method using liquid metal, the researchers achieved up to as high as 17 different metal elements mutually soluble HEA NPs (GaFeNiCuZnScVMnMgZrPtRhRuIrHfMoNb). In addition, GaPdWCuMg HEA NPs containing tungsten (W) with a high melting point (Fig. 3b) and GaCuCoCaNi HEA NPs with a large difference in atomic radius (Fig. 3c) were successfully prepared by this method. The researchers demonstrated that the elements in the two sets of HEA NPs are evenly distributed and have obvious periodic structures at the atomic level, with good crystallinity. These experimental pieces of evidence show that the synthesis of HEA NPs assisted by liquid metal can achieve the synthesis of HEA NPs with large differences in melting point and atomic radius. Therefore, it is proved that the liquid metal-assisted method has good universality.

Figure 3. Elemental distribution and structural characterization of liquid metal Ga-assisted synthesis of HEA-NPs
To verify the critical role of liquid metal in the synthesis of HEA NPs, the team simulated the synthesis process of HEA NPs by heating and venting hydrogen gas in an electron microscope chamber using an environmental electron microscope (ETEM). During heating from room temperature to 923 K, the Ga particles were first observed to flow and fuse with each other when the two Ga particles come close to each other (Fig. 4a). At the same time, under high temperature and hydrogen reduction atmosphere, metal nitrate precursor thermal decomposition into metal or metal oxides, metal oxides further reduced to metal elements. With the accumulation of Ga particles, the surrounding metal elements gradually mixed with Ga particles. When the temperature reaches 923 K, the original particles form two connected particles, one large and one small. After being held at 923 K for 30 minutes, the connected spheres split into two particles (Fig. 4b).
The change of surface tension of liquid Ga particles and the gas released by the metal oxide reduction reaction can explain the fusion-fission process as follows: In order to adapt to the change of surface tension, liquid Ga particles can be deformed to release strain; The gas produced during the decomposition and reduction of metal nitrates can also promote the movement of metal particles. During the alloying process, a large number of fusion and fission processes of Ga particles promote the mixing of various elements. After the in situ experiment, the samples were characterized by HAADF-STEM (Fig. 4c). The corresponding lattice fringes of two HEA NPs (Fig. 4c) show that the samples prepared in situ in ETEM present a good crystallinity with uniform distribution of elements, which is similar to that synthesized in the furnace. This is direct evidence of the formation mechanism of liquid metal Ga-assisted HEA NPs, which proves the feasibility of mass and high-quality preparation of HEA NPs in furnace tubes.
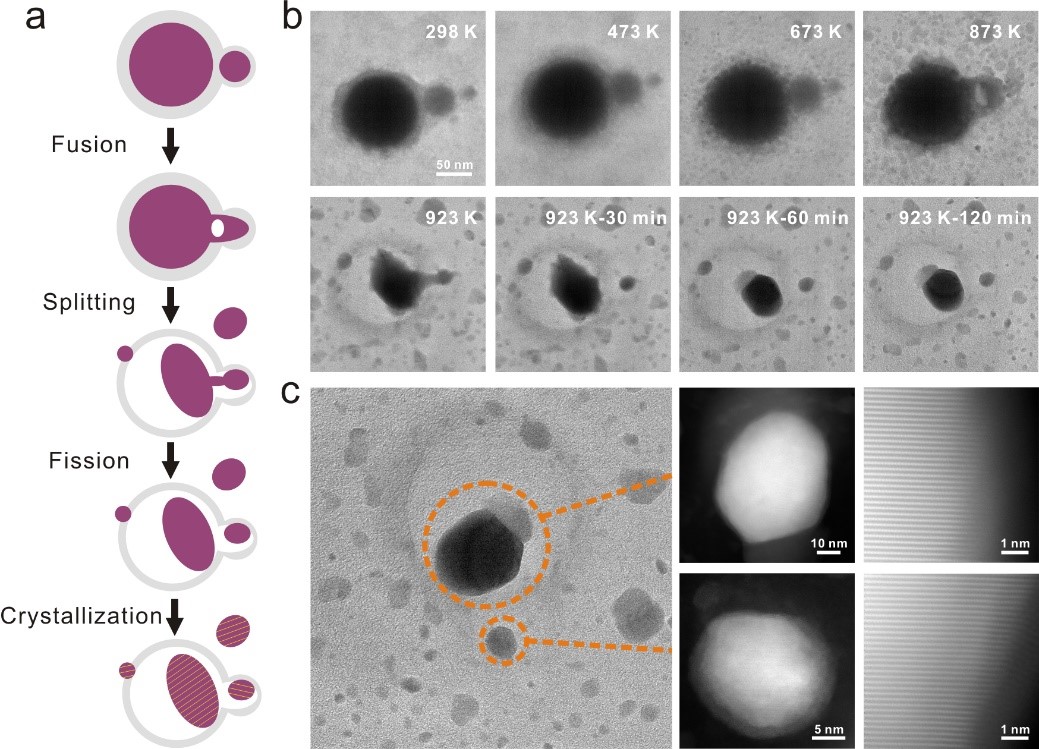
Figure 4. ETEM in situ observation and growth mechanism of liquid metal-assisted synthesis
In this work, a universal synthesis method of HEA NPs assisted by liquid metal was proposed, which showed unique advantages for the synthesis of HEA NPs with large differences in melting point and atomic radius. In situ transmission electron microscopy revealed the key role of liquid metal in the synthesis process of HEA NPs.
Guanghui Cao and Jingjing Liang, Ph.D. students from Wuhan University, Zenglong Guo, a Ph.D. student from the Department of Physics at SUSTech, and Kena Yang, a Ph.D. graduate of Wuhan University, are the co-first authors of this paper. Profs. Lei Fu, Mengqi Zeng, and Yuzheng Guo from Wuhan University, and Assoc. Prof. Junhao Lin from SUSTech are the co-corresponding authors.
Zenglong Guo studied the distribution of elements and the characterization of the in situ environment by electron microscopy. Gang Wang, a Ph.D. candidate from the Department of Physics at SUSTech, studied the structural characterization by atomic scanning transmission electron microscopy (STEM) as an important collaborator.
This work was supported by the National Natural Science Foundation of China (NSFC), Innovation and Entrepreneurship Team of Zhujiang River Talent Plan, High-Level Talent Team of Shenzhen, Key Laboratory of Shenzhen, and the Pico Center of SUSTech.
Paper link: